Why Antibiotics Are Failing Us—and What You Can Do Today to Fight Superbugs
Antibiotics once turned deadly infections into routine problems, but new WHO data show that a high percentage of common infections worldwide are no longer responding to our best drugs. This article explains what antimicrobial resistance is, why it’s rising so quickly, and the practical steps you and your community can take—at home, in clinics, and in policy—to help preserve the power of antibiotics.
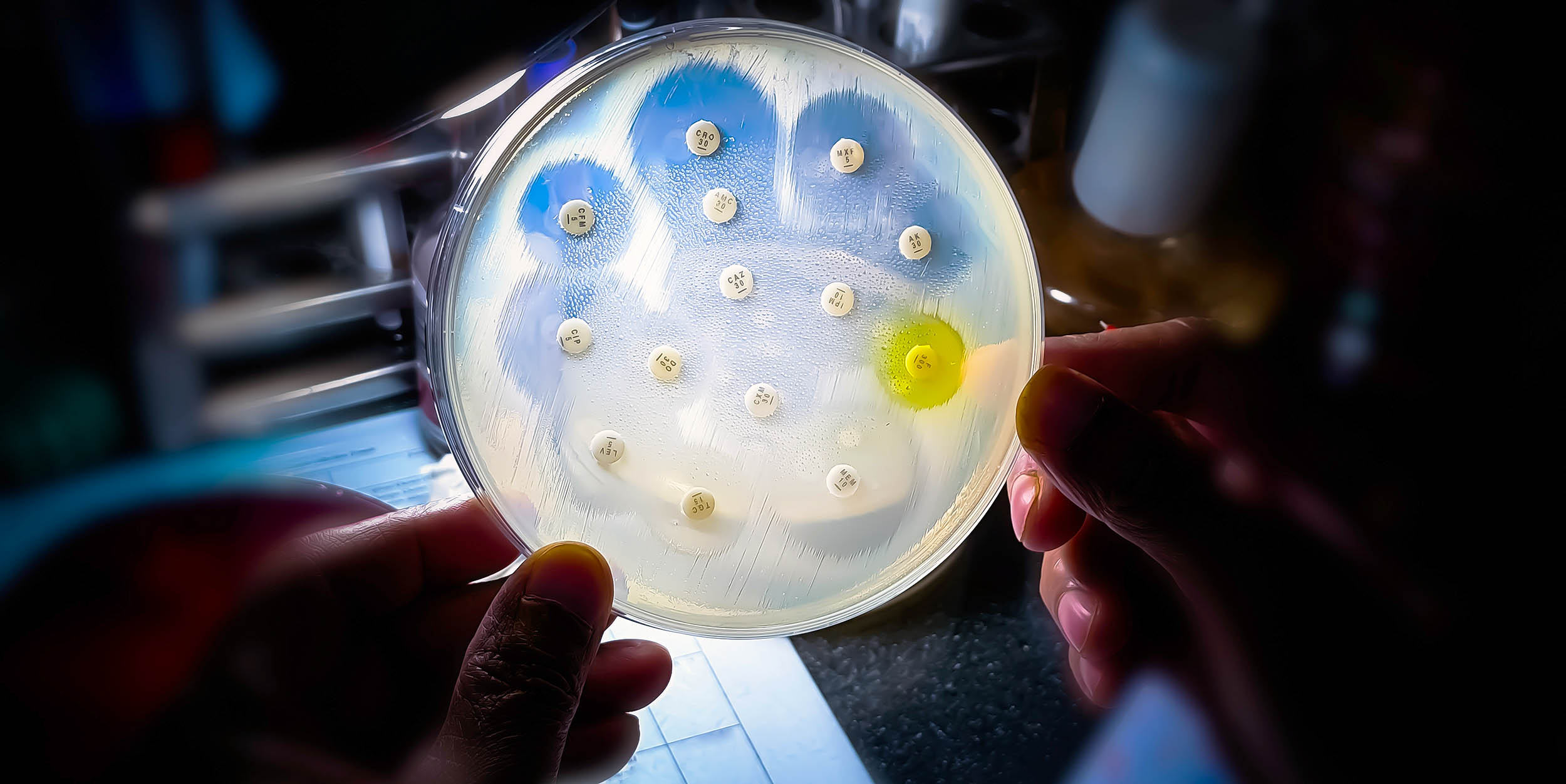
If you’ve ever taken antibiotics for a sinus infection or watched a loved one recover from pneumonia, you’ve experienced one of modern medicine’s greatest success stories. Yet, according to the latest World Health Organization (WHO) warning, that story is changing fast: a growing share of common infections—urinary tract infections, bloodstream infections, and sexually transmitted infections—are no longer responding to the antibiotics that used to work reliably.
This doesn’t mean antibiotics are useless. It does mean we’re entering a more fragile era, where simple infections can become dangerous again if we’re not careful. The goal of this guide is not to scare you, but to help you understand what’s happening and what you can realistically do—at home, as a patient, as a parent, or as a health professional—to protect yourself and others.
The WHO Warning: What’s Actually Happening With Antibiotic Resistance?
Antimicrobial resistance (AMR) happens when bacteria, viruses, fungi, or parasites evolve so that medicines designed to kill them no longer work effectively. The most urgent concern right now is antibiotic resistance in common bacteria.
In its latest surveillance updates, WHO reports that in many countries:
- A large share of Escherichia coli (E. coli) and Klebsiella pneumoniae infections are resistant to multiple first-line antibiotics.
- Some strains of gonorrhea are resistant to nearly all commonly used treatments.
- “Last-line” antibiotics—such as carbapenems and, in some cases, colistin—are losing effectiveness against certain hospital-acquired infections.
“We are already seeing situations where patients with common infections do not respond to available antibiotics. Without urgent action, we risk a future where routine surgery, childbirth, and cancer chemotherapy become far more dangerous.”
— Public health summary of WHO antimicrobial resistance briefings
Importantly, this is a global issue: high levels of resistance have been reported in both high‑income and low‑ and middle‑income countries. But the impact is felt locally—on your ward, in your clinic, in your family.
Why Antibiotic Resistance Matters to You (Even If You’re Healthy)
It’s easy to think of “superbugs” as a hospital problem or something that only affects very sick people. But antimicrobial resistance can affect anyone, at any age, in any country.
- Everyday infections become riskier.
A urinary tract infection (UTI) that used to clear with a 3‑day antibiotic course can now linger or progress to a kidney infection or sepsis if the bacteria are resistant. - Modern medicine depends on working antibiotics.
Cancer chemotherapy, organ transplants, joint replacements, and care for premature infants all rely on effective antibiotics to prevent and treat infections. - Costs and hospital stays increase.
Resistant infections often mean more expensive drugs, longer treatment, and longer time away from work or school. - Resistance can spread silently.
You can carry resistant bacteria in your gut or on your skin without symptoms and still spread them to vulnerable people.
The Science in Plain Language: How Bacteria Outsmart Antibiotics
Bacteria reproduce quickly. Each time they multiply, tiny genetic changes can appear. Most changes don’t matter, but occasionally a change helps a bacterium survive an antibiotic dose. When antibiotics are used:
- Susceptible bacteria die.
- Resistant bacteria survive and multiply.
- Resistance genes can spread to other bacteria—even across species—via mobile pieces of DNA.
The more often we use antibiotics, and the more inappropriately we use them (wrong drug, wrong dose, wrong duration, or for viral infections), the faster we select for resistant bacteria.

This is why WHO, the U.S. CDC, the European CDC, and many national health agencies all emphasize the same key message: we cannot “win” against resistance by drugs alone. We need a combined approach—prevention, smarter antibiotic use, and continued research.
What’s Driving the Rise in Antibiotic-Resistant Infections?
WHO’s latest warning highlights a familiar but powerful set of drivers. None of them act alone; together they create the “perfect storm” for resistance.
- Overuse in human medicine
Antibiotics are still frequently prescribed for viral infections (like colds, flu, most sore throats) where they don’t help, or given “just in case” without clear evidence of bacterial infection. - Misuse: wrong drug, dose, or duration
Skipping doses, stopping early, or using leftover antibiotics can expose bacteria to just enough drug to encourage resistance without fully clearing the infection. - Use in agriculture and animal husbandry
In many parts of the world, antibiotics are still used to promote growth or prevent disease in healthy animals, allowing resistant bacteria to spread through food, water, and the environment. - Infection prevention gaps
Overcrowded hospitals, inadequate sanitation, and limited access to clean water and vaccines all increase infections in the first place—driving higher antibiotic use. - Limited access to diagnostics
When rapid tests aren’t available, clinicians may feel pressured to prescribe broad‑spectrum antibiotics “just to be safe,” even if a narrower or no antibiotic would be better.
Practical Steps You Can Take to Help Fight Antibiotic Resistance
While governments and health systems must lead large‑scale action, your individual choices still matter. Think of antibiotic stewardship as a community effort: each person’s responsible use adds up.
1. Use Antibiotics Only When Truly Needed
- Don’t pressure clinicians for antibiotics if they explain they’re not needed.
- Ask, “Is this infection likely viral or bacterial?” and “What happens if we watch and wait?”
- Accept that sometimes the best care is supportive treatment (rest, fluids, pain relief) rather than a prescription.
2. Take Antibiotics Exactly as Prescribed
- Follow the dosing schedule carefully—set phone reminders if needed.
- Complete the full course unless your clinician specifically tells you to stop early.
- Never use leftover antibiotics or someone else’s prescription.
3. Prevent Infections Before They Start
The fewer infections we get, the less we need antibiotics. Evidence‑based prevention steps include:
- Staying up to date on vaccinations (e.g., flu, COVID‑19, pneumococcal, pertussis).
- Practicing regular handwashing with soap and water.
- Preparing and storing food safely to avoid foodborne illness.
- Using condoms and getting tested for sexually transmitted infections when appropriate.
4. Support Smarter Antibiotic Use in Animals and Food
- When possible, choose products from producers that commit to responsible antibiotic use.
- Handle raw meat carefully and cook thoroughly to reduce exposure to harmful bacteria.
- Support policies and retailers that phase out routine antibiotic use for growth promotion.
5. Advocate and Stay Informed
- Encourage local health facilities to join antibiotic stewardship programs.
- Stay updated with WHO, CDC, or your national health agency’s guidance on antimicrobial resistance.
- Share accurate information—not fear—within your community and on social media.
For Clinicians: Evidence-Based Stewardship in Daily Practice
If you work in healthcare, you’re on the front line of this challenge. Research on stewardship programs shows that thoughtful antibiotic use can reduce resistance and improve patient outcomes without increasing complications.
- Follow local and international guidelines.
Use up‑to‑date protocols based on local resistance patterns whenever possible. - Use diagnostics wisely.
Where available, use rapid tests and cultures to narrow treatment and de‑escalate broad‑spectrum drugs early. - Optimize dose and duration.
Shorter, guideline‑concordant courses are often as effective and safer than prolonged therapy. - Communicate clearly with patients.
Explain why antibiotics aren’t always needed and what red‑flag symptoms should prompt re‑evaluation. - Engage in or support stewardship programs.
Multidisciplinary teams (pharmacy, microbiology, infection prevention) consistently show benefit in reducing unnecessary use.
“Antimicrobial stewardship is not about restricting access—it’s about ensuring that every antibiotic we give has a clear indication, the right dose, and the right duration.”
— Infectious disease specialists’ consensus viewpoint
Before and After: How Stewardship Changes Real-World Outcomes
Hospitals and clinics that have implemented robust antibiotic stewardship programs offer a preview of what’s possible when we take resistance seriously.
- Before stewardship:
High rates of broad‑spectrum antibiotic use “just in case,” limited review of ongoing prescriptions, and growing rates of resistant infections and C. difficile. - After stewardship:
More targeted therapy, shorter courses, increased use of diagnostics, and measurable reductions in resistance trends and infection‑related complications.
Common Obstacles—and How to Overcome Them
Knowing what to do and actually doing it are different things. Here are some frequent barriers people describe, with realistic ways forward.
- “I feel worse and think only antibiotics will help.”
It’s natural to want fast relief. Ask your clinician what symptom‑relief options you have and what signs would indicate you need re‑evaluation. Many viral infections peak and then improve with time and supportive care. - “My patients expect a prescription.”
For clinicians: explaining your reasoning, offering a delayed prescription (to use only if symptoms worsen), or providing clear written advice can maintain trust while avoiding unnecessary antibiotics. - “Diagnostics aren’t available or affordable.”
This is a system‑level problem. Where tests are limited, use clinical scoring tools and established guidelines to reduce uncertainty, and advocate for better access to rapid diagnostics. - “Policy change feels slow and distant.”
Community‑level advocacy—through professional associations, patient groups, or local media—can help move antimicrobial resistance higher on the agenda.
The Bigger Picture: A One Health, Global Response
WHO and partner organizations emphasize a “One Health” approach: recognizing that human health, animal health, and the environment are deeply interconnected. Recent global reports stress:
- Coordinated surveillance of resistance in hospitals, communities, animals, and food.
- Investment in sanitation, vaccination, and infection prevention worldwide.
- Incentives for research into new antibiotics, diagnostics, and vaccines.
- International agreements to reduce unnecessary antibiotic use across all sectors.

While these high‑level efforts may feel distant from daily life, they are strengthened when individuals and professionals speak up and model good antibiotic practices in their own contexts.
Quick FAQ on Antibiotic Resistance
Do people become resistant to antibiotics?
It’s the bacteria, not your body, that become resistant. Your body doesn’t “get used to” antibiotics, but if you carry resistant bacteria, future infections may be harder to treat.
Is it ever okay to keep leftover antibiotics “just in case”?
No. Leftovers usually mean the course wasn’t completed as prescribed. Doses and durations should be tailored to specific infections; using leftovers can both harm you and fuel resistance. It’s safer to return unused medicines to a pharmacy take‑back program if available.
Can natural remedies replace antibiotics?
No home remedy has been proven to reliably cure serious bacterial infections the way antibiotics can. Supportive measures—like fluids, rest, saline nasal rinses, or honey for cough—can ease symptoms and are often appropriate for viral illnesses, but they are not substitutes for antibiotics when those are genuinely needed.
Evidence and Further Reading
For those who want to explore the science and policy in more depth, these authoritative sources provide up‑to‑date information on antimicrobial resistance:
- World Health Organization – Antimicrobial resistance (AMR)
- WHO – Global Antimicrobial Resistance and Use Surveillance System (GLASS)
- U.S. Centers for Disease Control and Prevention – Antibiotic / Antimicrobial Resistance
- European Centre for Disease Prevention and Control – Antimicrobial resistance
Moving Forward: A Realistic but Hopeful Outlook
Antibiotics changed the world—and they are still saving lives every day. The WHO’s latest warning is not a signal that all is lost; it’s a reminder that these powerful tools are finite and must be guarded carefully.
You don’t need to overhaul your life to make a difference. Choosing not to demand antibiotics for a viral illness, finishing a prescribed course correctly, washing your hands, getting vaccinated, or starting a stewardship initiative in your workplace—each step tilts the balance toward a safer future.

Today, choose one concrete action you can take: ask a question at your next clinic visit, review prescribing patterns in your department, or share a reliable resource with a friend. Small, steady choices, multiplied across millions of people, are exactly how we keep antibiotics working—for ourselves, and for the generations that follow.
